3-(Cyclohexylamino)-1-propanesulfinic Acid: More Than Just a Chemical Formula
Historical Development
In the world of sulfinic acids, 3-(Cyclohexylamino)-1-propanesulfinic acid stands out because of the path it has taken from lab curiosity to research staple. Early work around aminosulfinic acid derivatives kicked off in the mid-20th century, driven by a need to explore sulfur chemistry beyond the usual suspects like sulfonates or sulfates. Chemists looking for new ways to modulate chemical reactivity and biological activity found promise in sulfur, attached to an amino group and cyclohexyl ring. With advances in synthesis, researchers started controlling stereochemistry and purity better, opening the door for applications in chemical biology and organic synthesis that wouldn't have been possible a few decades back. These advances reflect not just technical skill but genuine curiosity and openness—two values that keep science moving forward.
Product Overview
3-(Cyclohexylamino)-1-propanesulfinic acid doesn't grab headlines like the big-name pharmaceuticals or cutting-edge catalysts, but it chips away steadily at problems chemists want to solve. At its core, the compound merges a cyclohexylamino group with a sulfinic acid tail, creating opportunities for unique chemical reactivity. Its structure offers a rare combination: the stability that comes from a saturated cyclohexyl ring and the chemical versatility provided by the sulfinic acid group. This allows researchers to use it as both a building block and as a functional modulator. Over the years, labs have leaned on it when designing molecules that require tailored reduction or radical scavenging properties—tasks with real-world impact in drug and catalyst development.
Physical & Chemical Properties
Chemicals like 3-(Cyclohexylamino)-1-propanesulfinic acid rarely announce their usefulness at first glance. The white-to-off-white crystalline solid points toward good stability, but the crucial action happens elsewhere. The presence of the sulfinic acid group leads to interesting acid-base chemistry, letting this compound engage in both nucleophilic and electrophilic reactions. Water solubility is a challenge due to the hydrophobic cyclohexyl group, leading some folks in the lab to rely on organic solvents for handling. When heating, the compound holds up to moderate temperatures without decomposing, a boon for reactions that require a little push. The balance between hydrophobic and hydrophilic parts of the molecule keeps it in a sweet spot for chemists looking to explore new reactivities.
Technical Specifications & Labeling
There’s plenty of jargon out there, but clarity matters more in the lab than a fancy label. Purity—often above 98 percent—usually makes the difference between a successful experiment and a frustrating repeat. The compound featured under several CAS numbers and synonyms, depending on supplier and region. Researchers know from experience how tracking down a synonym can mean the difference between a smooth procurement process and a wasted afternoon with outdated catalogs. Good labeling not only acts as reassurance for safety but builds trust from scientists who want to avoid contamination and mishaps. Those details help everyone upstream and downstream, from synthesis bench to publication.
Preparation Method
Preparing 3-(Cyclohexylamino)-1-propanesulfinic acid, for most labs, stays grounded in classical organic synthesis. One common approach starts from cyclohexylamine, passed through a Michael addition to acrylonitrile, yielding the key intermediate. Reductive processes follow, and then careful oxidation introduces the sulfinic acid group. Yields depend on attention to reaction conditions—too much oxidation, and side reactions dominate, erasing hours of work. As with most organic syntheses, patience pays off. Crystallization helps purify the product, and the right solvent system makes the final separation much less of a gamble.
Chemical Reactions & Modifications
This compound steps up when a reaction needs a selective reduction, scavenging radicals, or modifying certain electrophilic centers. The sulfinic acid group’s nucleophilicity allows alkylation or acylation, letting researchers stitch it into larger frameworks or small-molecule analogs. The cyclohexylamino portion isn’t just window dressing; it moderates basicity and steric profile, so this sulfinic acid finds favor in fine-tuned syntheses. People in medicinal chemistry have pulled this structural motif into lead compound optimization, where subtle changes to a molecule's interaction with biological targets can mean a world of difference.
Synonyms & Product Names
Few chemicals travel under a single name. 3-(Cyclohexylamino)-1-propanesulfinic acid appears in lab notebooks as N-cyclohexyl-3-aminopropane-1-sulfinic acid, cyclohexylaminopropanesulfinic acid, and various catalog-jeopardy renditions. This mess of names reflects how chemists from different backgrounds or suppliers approach the molecule. The result: scientific articles and procurement forms benefit from careful double-checking, since the paper chase for synonyms reroutes projects more often than one might expect. Save yourself a hassle—always cross-reference before signing off on an order or submitting a grant proposal.
Safety & Operational Standards
Handling any sulfinic acid calls for respect, not just because of what it could do to your skin but because unplanned reactions can mess up a lab bench in minutes. Gloves, eye protection, and a working fume hood stay mandatory, especially since the compound can release irritant gases under heat or during side reactions. It’s easy to cut corners in a rush, but experience teaches that a minor slip often costs days lost to recovery and paperwork. Disposal of sulfinic acids must follow local regulation, not only to keep water sources safe but to avoid hefty fines and public scrutiny. Labs that maintain good logs and training sessions manage to avoid those all-too-common close calls.
Application Area
This compound’s story mostly unfolds in research institutions and pharmaceutical labs. Organic chemists take advantage of its ability to modify molecules through selective sulfinylation or deprotection. Biologists, on the other hand, scrutinize its radical scavenging, digging into its use as a model antioxidant. In specialized cases, chemical engineers work with it as an additive during processing steps needing sulfur-based stabilization. These practical uses might not make the evening news, but incremental improvements in molecular synthesis and antioxidation often feed bigger breakthroughs days or months down the line. My own experience drives home how a reliable reagent can quietly tip the scales toward new discoveries.
Research & Development
Ongoing research taps this compound for its versatility. Teams focus on expanding its utility in green chemistry applications, exploring catalysts based on the sulfur center and probing deeper into its reactivity under mild conditions. Computational chemists use its structure to calibrate models predicting sulfinic acid reactivity, feeding into machine learning approaches that design new molecules faster than ever before. As industry shifts towards sustainability and waste reduction, reactions leveraging the mild and selective features of sulfinic acids gather interest. Whether for pharmaceuticals, agrochemicals, or specialty polymers, incremental refinement continues—people look for less waste, higher yield, and safer processes.
Toxicity Research
Every novel compound raises concerns about toxicity. Data around 3-(Cyclohexylamino)-1-propanesulfinic acid remains limited, which is all the more reason labs monitor exposures closely. Standard animal model studies aim to illuminate short- and long-term effects, tracking both direct toxicity and environmental persistence. That diligence reflects not just regulatory obligation, but an ethic of care around lab safety. No one benefits from shortcuts on toxicity research, especially when a compound could find its way into processes scaling from grams to kilograms. It’s responsible to keep updating hazard evaluation, not just relying on what's already published.
Future Prospects
Compared to elements like carbon or nitrogen, sulfur’s full chemistry stays underexplored, which means 3-(Cyclohexylamino)-1-propanesulfinic acid won’t be going out of style anytime soon. The future likely brings refinements in both its preparation and application, guided by automation and high-throughput screening. As more industries press for greener alternatives, this acid and its derivatives could step up either as direct reagents or as reference points for designing more sustainable solutions. Younger chemists and seasoned researchers alike pay attention to where new cross-couplings, medicinal chemistry tweaks, or bioactive molecule modifications become easier because of this one molecule. It has all the makings of a sleeper hit—helpful in the background, with its real value becoming clearer as bigger problems find creative solutions in chemistry’s toolbox.
Getting to the Heart of the Molecule
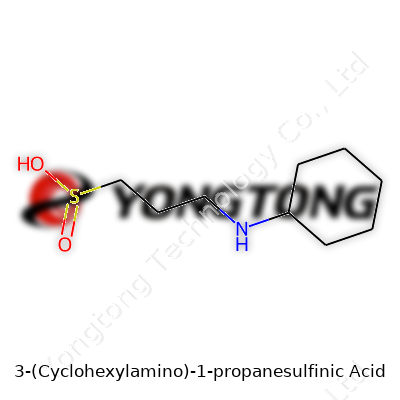
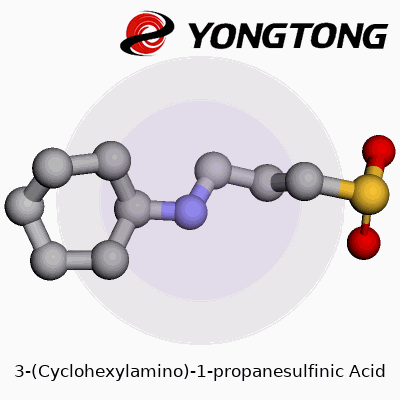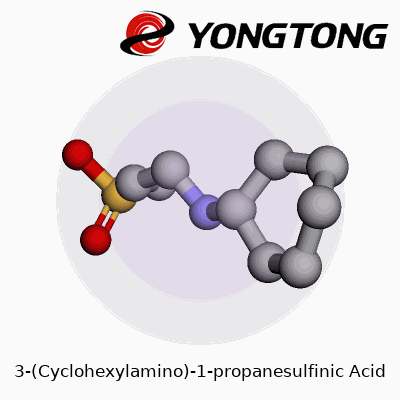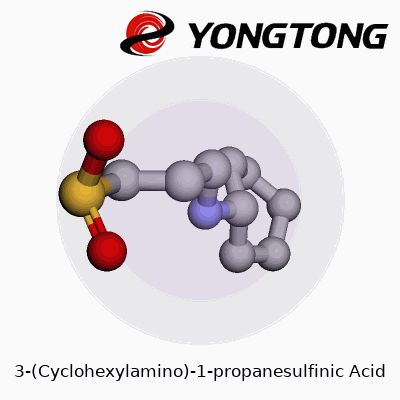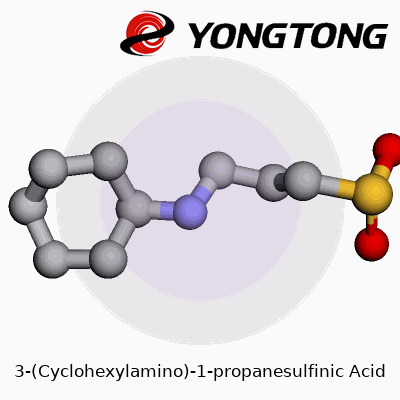
Understanding a compound like 3-(Cyclohexylamino)-1-propanesulfinic acid means breaking it down to its skeleton. The molecule features a cyclohexyl ring, which is a six-carbon ring shaped in the classic ‘chair’ form seen in organic chemistry classrooms. Attached to that is an amino group, which often acts as a reactive site in chemical reactions. The rest of its backbone, a three-carbon chain ending in a sulfinic acid group, brings a unique set of properties. Put these pieces together and you meet a molecule that offers much more than just a jumble of atoms.
Why the Structure Calls for Attention
In labs and pharmaceutical companies, structure often decides function. A cyclohexyl ring lends bulk and hydrophobic character, shielding parts of the molecule from water and influencing how the compound interacts with enzymes or cellular targets. Adding an amino group isn’t just for show; it provides a ‘handle’ for linking up with other chemical entities or biological molecules. The sulfinic acid feature brings reactivity that you won’t find in more common sulfonic or carboxylic acids. The sulfur atom, sitting in an intermediate oxidation state, brings a special chemistry — for instance, it acts as both a nucleophile and a mild oxidant, something medicinal chemists find useful.
Real-World Applications and Relevance
Medicinal chemists and researchers often chase after molecules with this sort of architecture. A compound like 3-(Cyclohexylamino)-1-propanesulfinic acid fits perfectly into enzyme pockets that prefer some bulk, but also want a precise chemical ‘pin’ to hold on to. Novel antibiotics, cancer drugs, and enzyme inhibitors sometimes rely on these properties. The chemical structure’s balance of rigidity, flexibility, and reactivity lets researchers fine-tune how a molecule will behave. Drugs that only work in specific environments or need to be ‘switched on’ at the right time start with building blocks like this.
Concerns and Considerations
Chemists don’t just draw molecules on paper for fun. Lab synthesis, toxicity, metabolic stability — these issues show up as soon as a molecule leaves the drawing board. The cyclohexyl ring often survives metabolic attack longer than straight-chain carbon groups, making it attractive for drug development. At the same time, sulfinic acid groups come with their own metabolic quirks. They can act as redox agents in the body, which can be both helpful and harmful depending on the context. Getting the benefits without the backlash takes skill and knowledge grounded in both chemistry and biology.
Paths Forward
If researchers want to squeeze the most out of compounds like this, collaboration remains the best route. Chemists, toxicologists, and clinicians each bring expertise that makes the whole effort safer and more productive. Better predictive models could help judge metabolism and toxicity before a single pill sees human trials. Chemists know that making small tweaks to a molecule’s blueprint can change its story in huge ways. Staying open to that kind of thinking, and combining it with solid experimental evidence, will keep useful compounds coming out of the lab and making a difference in the real world.
A Quiet Workhorse in Modern Lab Benches
No one brags about the sulfinic acids hiding in test tubes, but if you look at synthetic chemistry labs, you’ll find 3-(Cyclohexylamino)-1-propanesulfinic acid making a solid contribution. Having spent a chunk of my career shuffling between chemical benches, I can say that this compound earns its keep in a surprising number of research corners.
Improving the Toughest Reactions
Organic chemists chase efficient ways to make bonds that nature doesn’t hand out for free. Sulfinic acids, and especially this one, bring an edge to several reactions by acting as reducing agents. They help shuffle electrons around, turning mystical lab transformations into repeatable steps. These aren’t small-time tasks: pharmaceuticals depend on reliable reactions to make safer, purer medicines. A compound like this one ends up streamlining the tricky reduction of nitro groups, sulfonylations, and other transformations. I’ve watched postdocs celebrate smooth results using it when more brute-force methods fell short.
Clever Applications in Drug Discovery
The early stages of drug discovery fight with tough molecules. Nature’s complexity means chemists have to work with stubborn functional groups. 3-(Cyclohexylamino)-1-propanesulfinic acid slips into the mix with enough reactivity to enable selective modifications without trashing delicate parts of the molecule. As drug development pushes for innovative scaffolds and links, this sulfinic acid’s role as a transfer reagent stands out. It helps introduce or swap out building blocks, letting scientists build up the molecules under less hazardous conditions.
By making life a bit easier for medicinal chemists, it can shave weeks or months off a project timeline—not just saving money, but sometimes speeding up access to a life-changing therapy. That’s not pie-in-the-sky talk; the “fail fast, iterate fast” approach leans on such practical advances.
Helping Manufacturers Skip a Few Steps
Scale-up teams looking for cost savings don’t overlook the details. Bulk chemistry favors processes that cut down the number of steps or use milder conditions, and this is where 3-(Cyclohexylamino)-1-propanesulfinic acid enters the strategy session. Companies look for reagents that let them avoid expensive catalysts, and this compound can behave predictably under a range of conditions. For anyone tracking the supply chain carbon footprint, every shortcut counts.
Environmental Questions and Responsible Practices
Every chemical actor brings environmental tradeoffs to the script. The use of sulfinic acids in industry puts pressure on waste treatment protocols. Factories have to dispose of byproducts without dumping pollutants into groundwater. Future-thinking researchers are exploring greener analogs or recycling strategies, but in the meantime, stricter regulations and careful engineering keep this chemistry productive without a heavy environmental toll.
Lab Results That Make the News
Very few people outside a research lab ever hear about intermediates like 3-(Cyclohexylamino)-1-propanesulfinic acid. Yet, its impact quietly ripples into public health, cleaner manufacturing, and science driven by real-world needs. Highlighting its work helps explain why funding basic chemical research matters—the breakthroughs depend on the building blocks and the clever hands assembling them.
Why Proper Chemical Storage Hits Home
I’ve watched chemists pop open sample containers as if they were pulling snacks from a pantry. Years later, I learned tough lessons from burnt fingertips and breathing trouble—things only told by those who know what a slip can cost. Chemicals like 3-(Cyclohexylamino)-1-propanesulfinic acid demand serious respect at every step. Even if a compound seems stable at room temperature, mistakes in storage or handling reveal risks that affect not only labs, but anyone who’s downwind from that research building.
Simple Storage, Serious Standards
Every science classroom posts the “cool, dry, well-ventilated” rules for storing chemicals. Turns out, nothing beats that advice. This compound doesn’t fall into the explosive or volatile hall-of-fame—at least from what research shows—but I’ve seen powder and crystalline materials clump up or degrade within weeks if left in humid corners or poorly sealed jars. Moisture, direct sunlight, and heat all chip away at chemical stability, sometimes transforming something useful into something dangerous.
Most labs use polyethylene or glass bottles with tight seals for storage. Forgetting a label or mixing up container lids creates confusion, and that’s often where accidents sneak in. On more than one occasion, even professionals confuse two similar-looking substances, which raises the stakes for accurate labeling and organized shelving.
PPE Is Non-Negotiable
I’ve known colleagues who refused to glove up, brushing off the risk because “they’re careful.” That lasts until a cut forms, or skin gets irritated. 3-(Cyclohexylamino)-1-propanesulfinic acid falls into the category where gloves, lab coats, and goggles aren’t up for debate. Powders drift in air, then land on hands, faces, or clothing without warning. Even tiny airborne particles, once inhaled or rubbed into eyes, bring a world of regret.
Ventilation turns into a mainline defense. Fume hoods aren’t just a suggestion—they protect not only the user, but everyone else in that room. Without them, one person’s mistake becomes everyone’s problem.
Cleaning Up and Waste Matters
Waste piles up fast. I remember a project where containers of used chemical sat out for weeks. The smell eventually revealed the problem. It’s never enough to stick leftovers in a sink and forget it—solids, liquid residues, or spills get sent off for professional disposal. Dilution in a sink or tossing in regular trash breaks both common sense and local laws. From my experience, good habits start with a clear waste protocol, shared up front and enforced daily.
Keeping Training Routine—and Real
Seeing a new team member treat everything like table salt makes me nervous. Regular, straightforward training helps people understand the risks before they get hurt. Stories from past mistakes stick much better than dense manuals. I’ve seen labs run drills on what to do if something goes wrong, which eliminates panic later.
Moving Forward—One Good Practice at a Time
At the end of every shift, it’s the basics that save the most headaches: dry, labeled containers; fresh gloves; clean, shielded benches. Respect for chemicals comes from experience and from watching others pay for small mistakes. Keeping each other honest and implementing solid routines create the safety net that protects a whole team, whether the compound is notorious or quietly potent.
Purity Looks Simple, But It’s Not Just a Number
Most people only start thinking about purity when a product isn’t working as they expected, or worse, turns out to be unsafe. Years of working with everything from cleaning agents to baking ingredients taught me that not all “pure” products are created equal. For example, a label boasting 99.9% purity might sound impressive, but questions should pop up: What’s in the missing 0.1%? Are those trace elements harmless, or could they end up spoiling a batch or causing damage over time? In research and food, impurities at these tiny levels can still cause noticeable problems. The higher the purity, the more reliable the result, whether you’re mixing chemicals in a lab or making sourdough bread at home.
Shoppers placing trust in purity figures need to remember that no strong regulatory body oversees every category of product. In cleaning products and minerals, manufacturers sometimes inflate claims, hoping people won’t check details. But the stakes are high: unwanted additives or contaminants in industrial chemicals can damage equipment, while poorly filtered table salt may add unwanted flavors or health risks. A modest extra investment in something genuinely pure saves headaches in the long run. That’s been my experience, both as a buyer and as someone who’s had to troubleshoot mysterious failures in kitchens and home projects.
Packaging Size Shapes Experience—And Waste
Packaging size rarely gets the spotlight, but it can make or break how practical a product is. In professional kitchens and factories, bulk sizes offer cost savings and reduce the hassle of running out mid-job. Yet, opening a big bag at home or in a small lab turns into a race against time—humidity, air, and sunlight degrade content faster than most people realize. Splitting a large container into smaller, airtight jars at home helps, but now you’re wrestling with extra containers and keeping careful notes on dates, which often doesn’t happen.
I learned quick lessons on this front running a small soap-making shop. Ordering 25-kilo sacks seemed thrifty. They saved money, but powder grew clumpy even on cool, dry days, leading to wasted product. Friends in the restaurant business echoed similar complaints with spices. Smaller, well-sealed packs extend shelf life, ease storage, and produce less waste, especially for home users or small business owners who don’t spend ingredients quickly. Manufacturers sometimes ignore this, sending everything in industrial-sized tubs, yet the people using them often need the exact opposite: smaller, fresher, less cluttered.
How We Fix It
Companies benefit when they move away from a “one size fits all” mindset. They can build trust with clear purity disclosures, not just vague claims. This matters for communities with allergies, for scientists with experiments on the line, and for anyone who cares about the hidden contents in their pantry or workshop. Smart brands also offer options for packaging size, giving customers power to buy only what they’ll actually use.
Regulators and watchdogs could hold manufacturers accountable for everything they put on labels. Random audits and public reporting—already common in some countries—shine a spotlight on overstated claims or misleading packaging. On the consumer side, asking questions, searching for certifications or independent lab tests, and sharing experiences with others in forums drives companies toward transparency and integrity. This might seem small, but every accurate label and right-sized package makes life easier and safer for all of us.
Why Chemical Safety Demands Attention
In any lab or industrial space, nothing feels as routine as handling chemicals — until someone winds up with a burn, respiratory issue, or a spill that empties a room. Working with lesser-known compounds like 3-(Cyclohexylamino)-1-propanesulfinic acid can fool even experienced hands, since the risks might not be as well publicized as with stronger acids or volatile solvents.
Over the years, I’ve seen labs where solid rules make a difference and others where shortcuts become the norm. The quiet truth: many hazards don’t grab headlines until damage is done. Acids have a way of finding any weak spot — a worn glove, carelessly wiped surface, or a mask worn too loose.
Skin, Eyes, and Breath: Entry Points for Danger
The structure of 3-(Cyclohexylamino)-1-propanesulfinic acid hints at trouble. Many sulfinic acids irritate skin and eyes, and this compound’s amine group adds more bite. Direct contact usually leads to immediate discomfort — redness, itching, swelling. Protective gloves rated for acids keep the odds in your favor, but even the best gloves can tear without you noticing.
Splashes are every lab worker’s worry. No one likes acid in the eye. Goggles, not glasses, matter. In a crowded chemistry course, I once saw a student ignore splash-proof goggles for a “quick transfer.” The resulting trip to the safety shower triggered weeks of paperwork.
Fumes present a less obvious threat. Even when a compound looks solid and smells faint or not at all, it can irritate lungs or cause headaches. Decent ventilation goes a long way, and using a fume hood means fewer air quality complaints and fewer late-night coughs.
What Happens if You Take Shortcuts
Nobody sets out planning for an accident, but people get sloppy. Mixing incompatible chemicals, skipping lab coats, or storing something incorrectly turns a safe day chaotic. Sulfinic acids often react with oxidizers, metals, or strong bases. That means you’re one mistake away from dangerous gases or uncontrolled heat. In my early days, I watched a flask bubble over because someone rinsed it out with bleach instead of following the chemical waste guide. The reaction ruined the day, and could have been worse if we didn’t get everyone out fast.
Waste and Storage: Real-World Concerns
Dumping chemicals down the drain or tossing them in regular trash no longer flies. The push for lab sustainability forced everyone to look at waste streams and double-check storage. Sulfinic acids can degrade, and their decay may release more hazardous byproducts. Keeping them in airtight, clearly-labeled containers, tucked away from sunlight and incompatible materials, keeps everyone safer and keeps the regulators happy.
Accurate labeling might seem like bureaucratic nonsense, but it saves time in an emergency. A colleague once opened a vial, expecting a harmless buffer, and almost ruined her lunch break with the fumes. Good labels stopped things from getting worse.
Building a Culture of Safety
Working with unfamiliar chemicals, communication makes a real difference. Training, regular risk reviews, and treating even “mild” acids with caution changes outcomes. Keeping emergency showers and eyewash stations unobstructed, running drills that don’t feel pointless, and sharing stories of close calls or mistakes can turn rules into habits.
Laboratories can’t remove all risk from chemicals like 3-(Cyclohexylamino)-1-propanesulfinic acid, but active awareness and stubborn attention to detail help dodge most dangers. Chemical accidents aren’t always flashy, but they leave marks — on equipment, on people, and in the records. Practicing good habits holds the line when warnings and theoretical guidelines fade into routine.